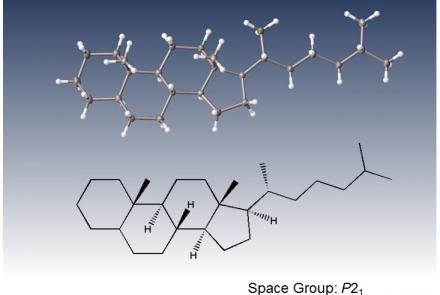
XtaLAB Synergy-S
Ventajas
- Recolección de datos más rápida y precisa debido al goniómetro kappa de alta velocidad, la fuente de rayos X de alto flujo, el detector de rayos X rápido y de bajo ruido, y el software de control de instrumentos altamente optimizado.
- Versatilidad experimental mejorada cuando la opción de fuente dual se selecciona entre tres posibles longitudes de onda (Mo, Cu o Ag).
- El más alto nivel de seguridad del usuario con múltiples circuitos de seguridad electromecánicos redundantes integrados en la cámara de radiación diseñado ergonómicamente.
- Minimice su tiempo de inactividad utilizando diagnósticos en línea integrados y solución de problemas para diagnosticar y solucionar casi todos los problemas sin visitar el sitio.
- Resuelva estructuras automáticamente y determine su muestra en unos segundos antes de comprometerse con un conjunto de datos completo mediante el uso de la característica “What is this?” (¿Qué es esto?).
- Mejore su capacidad de investigar muestras pequeñas porque la tecnología de píxeles de matriz de estado sólido de los detectores de rayos X HyPix, demuestran que los fotones de rayos X se cuentan instantáneamente a medida que llegan al detector. No hay conversión a luz visible por un centelleador, por lo que la energía del fotón se puede evaluar en el momento de la detección, lo que genera imágenes esencialmente libres de ruido. Las imágenes libres de ruido demuestran que puede contar con más tiempo para los cristales de difracción débil, sin pérdida en la calidad de los datos derivada del ruido del detector.
- Optimice la velocidad de recolección de datos cuando seleccione el detector curvo opcional HyPix-Arc 150°, que permite una cobertura theta que excede los detectores más grandes y al mismo tiempo ofrece la tecnología de detección de mayor rendimiento.
- Mejore su capacidad para resolver celdas de unidades grandes, redes inconmensurables o duplicadas cuando selecciona la ranura de haz variable motorizada opcional, para alterar la divergencia y adaptar la fuente a los requisitos de su muestra.
Lea lo que dicen nuestros clientes sobre XtaLAB Synergy-S
Difractómetro de rayos X de microfoco único o doble para todas sus necesidades de cristalografía
Un difractómetro de rayos X de monocristal rápido y ágil para el análisis de estructuras 3D de moléculas pequeñas
Especificaciones y características
| Nombre del producto | XtaLAB Synergy-S |
| Atributos principales | Difractómetro de fuente de rayos X de microfoco de tubo sellado único o doble con detector híbrido de píxeles de matriz y goniómetro kappa |
| Detectores | HyPix-6000HE o la amplia cobertura theta opcional HyPix-Arc 100° o HyPix-Arc 150° |
| Fuente de rayos X | Fuente de rayos X PhotonJet-S con un nuevo microfoco de tubo sellado que incorpora un nuevo diseño de espejo y un nuevo hardware de alineación. Hay tres tipos de objetivos disponibles (Mo, Cu, Ag) |
| Goniómetro | Rápido goniómetro de geometría kappa que permite velocidades de escaneo de recolección de datos de hasta 10°/seg |
| Accesorios | Oxford Cryostream 800, Oxford Cobra, XtaLAB Synergy FLOW robotic system, XtalCheck-S, High Pressure Kit |
| Computadora | PC externa, MS Windows® OS |
| Dimensiones principales | 1300 (ancho) x 1875 (alto) x 850 (profundo) mm |
| Cuerpo | 550 kg (unidad central) |
| Requerimientos de energía | 1Ø, 90-130 V 15 A o 180-260 V 4 A |